| biochem_and_cells_review.pdf |
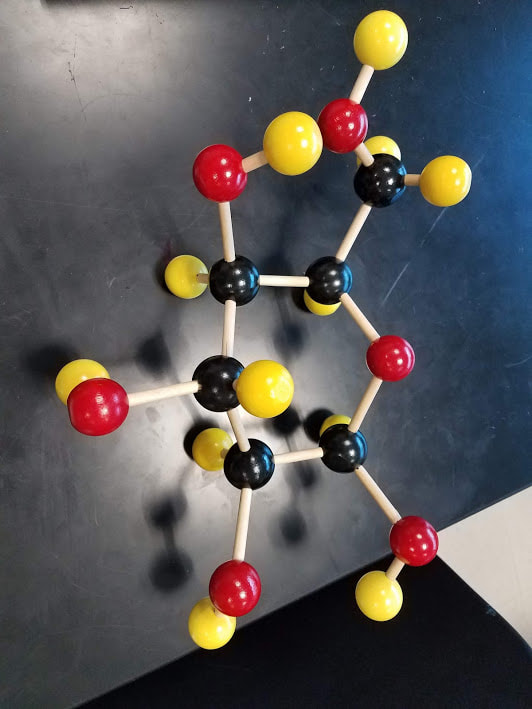
 We practiced testing for different nutrients using our indicator solutions. We used iodine to test for the presence of starch in a piece of apple and in a piece of roll. When the amber colored iodine was added to the apple, it remained amber, this indicated that starch was NOT present. When the amber colored iodine was added to the roll it turned black, this indicated that starch was present. Next we used Benedict's solution to test for the presence of glucose in a piece of apple as well as in a starch solution. When using Benedict's solution as an indicator, it must be heated to show the results. When the blue colored Benedict's solution was added to the apple and heated, it turned brick orange, this indicated that glucose was present. When the blue colored Benedict's solution was added to the starch solution and heated, it remained blue, this indicated that glucose was NOT present. Finally we used Beirut solution to test for the presence of protein in milk. We added the blue Beirut solution to milk in a test tube and it changed to a purple color indicating that protein was present.  Getting used to building molecular models. Ethane, Mehtane and Octane. On paper the formula for octane is written out in a straight line. Due to the angle at which bonds forms in the real world it looks more like a zig-zag shape.  Some molecules form double or triple bonds. We used multiple springs to represent this same idea in our molecular models. This is a rather accurate depiction of the molecules. Above is a 3D molecular model of glucose constructed by the students in class. We then demonstrated the process of dehydration synthesis (removing water to make something bigger exist) to join together two glucose molecules to make a molecule of maltose. We then demonstrated hydrolysis or digestion (adding water to break it back down) to break the molecule of maltose back down to two molecules of glucose.
All organic compounds contain a carbon atom bonded to a hydrogen atom.
Organic Molecule Examples
Inorganic Molecule Examples
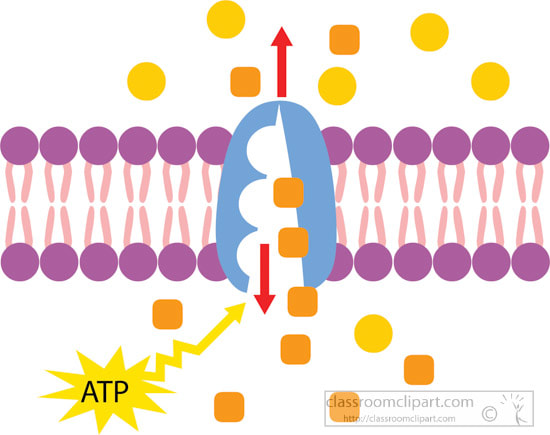
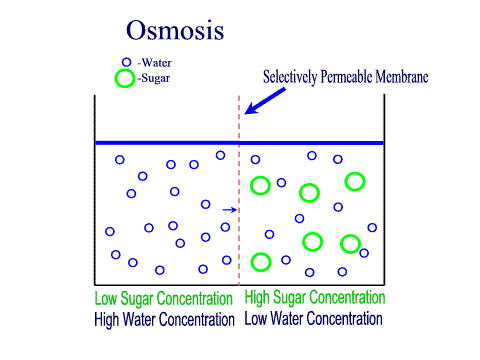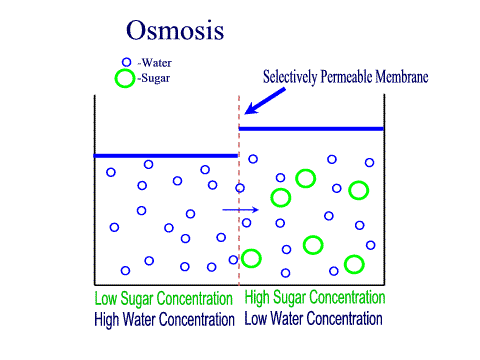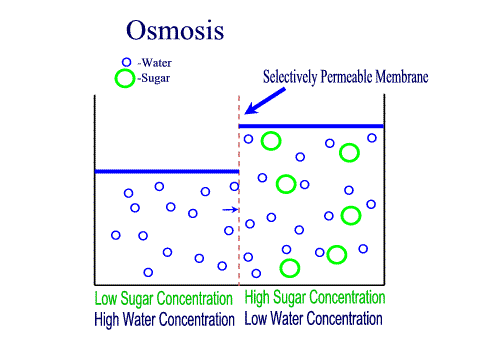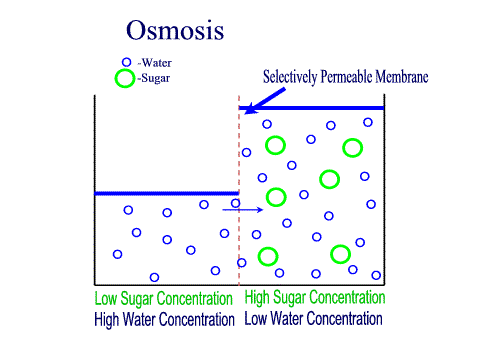
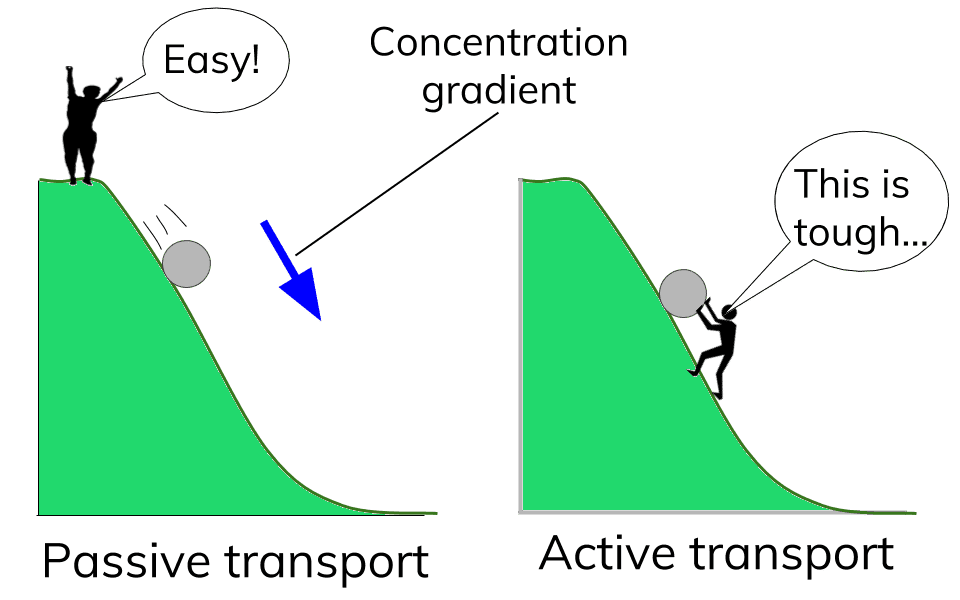
Carbohydrates Simple sugars such as glucose are the building blocks of carbohydrates. Carbohydrates are large molecules that provide energy for the life processes through the process of cellular respiration which occurs in the mitochondria of cells. Protein Amino acids are the building blocks of proteins (at least 50 amino acids are needed to form a protein). Proteins are large molecules that are needed for cell growth and repair. Enzymes (a type of protein) play a vital role in cell function. Lipids Glycerol and Fatty acids are the building blocks of lipids. Lipids are generally our fats, waxes and oils. They provide us with insulation, protection and energy reserves. Nucleic Acids DNA and RNA contain hereditary information. DNA is like the recipe book for life. DiffusionMolecules will naturally flow from an area of high concentration to an area of low concentration without any energy Active TransportThe movement of molecule from an area of low concentration to an area of high concentration, this requires energy (ATP) OsmosisThe diffusion of water across a selectively or semi permeable membrane. Water molecule flow until equilibrium is reached. Passive vs Active transportPassive is like rolling down a hill, high to low, requires no energy
Active is like walking back up the hill, low to high and requires a lot of energy The cell membrane is also known as the plasma membrane. It is a membrane that separates the interior of all cells from the outside environment which protects the cell from its environment. The cell membrane consists of a lipid bilayer with embedded proteins. The cell membrane controls the movement of substances in and out of cells and organelles. In this way, it is selectively permeable. Safety Measurement and MetricsTools and Techniques of the Biologist GraphingScientific Method Organization of Life Life FunctionsCell Basics Review Sheet with Answers
|
Ms. JonesLiving Environment 2019-2020 |
||||||||||||